Apply online for service right now. Our experts are glad to help you with the solution and by all means will answer all your question
Expert holding "SOYUZEKSPERTIZA" of the RF CCI has developed a special line of services for companies and organizations of the Healthcare sector. We are glad to offer the full range of services for securing permit licenses for circulation of domestic and foreign medical devices in Russia. We carry out comprehensive consulting support for launching medical devices into Russian healthcare market. Cooperation with us also allows companies to gain access to the public procurement market.
According to Federal Law No. 323-FZ dd. November 21, 2011 “On Basics of Health Protection of Citizens in the Russian Federation” all medical devices are subject to compulsory registration. A document confirming state registration and entering a medical device in the State Register is a Registration Certificate of Roszdravnadzor.

As part of the registration process, our company provides:
- full audit of documents required for registration of medical devices, assistance in obtaining missing information;
- registration of a Permit license for import of testing samples of medical devices for state registration;
- preparing a set of technical documentation in accordance with the Order of the Ministry of Healthcare of Russia No. 11n
- dd. 19.01.2017;
- organization of necessary tests in reliable testing centers certified by Roszdravnadzor;
- monitoring of each stage of the registration process.
order a free request processing
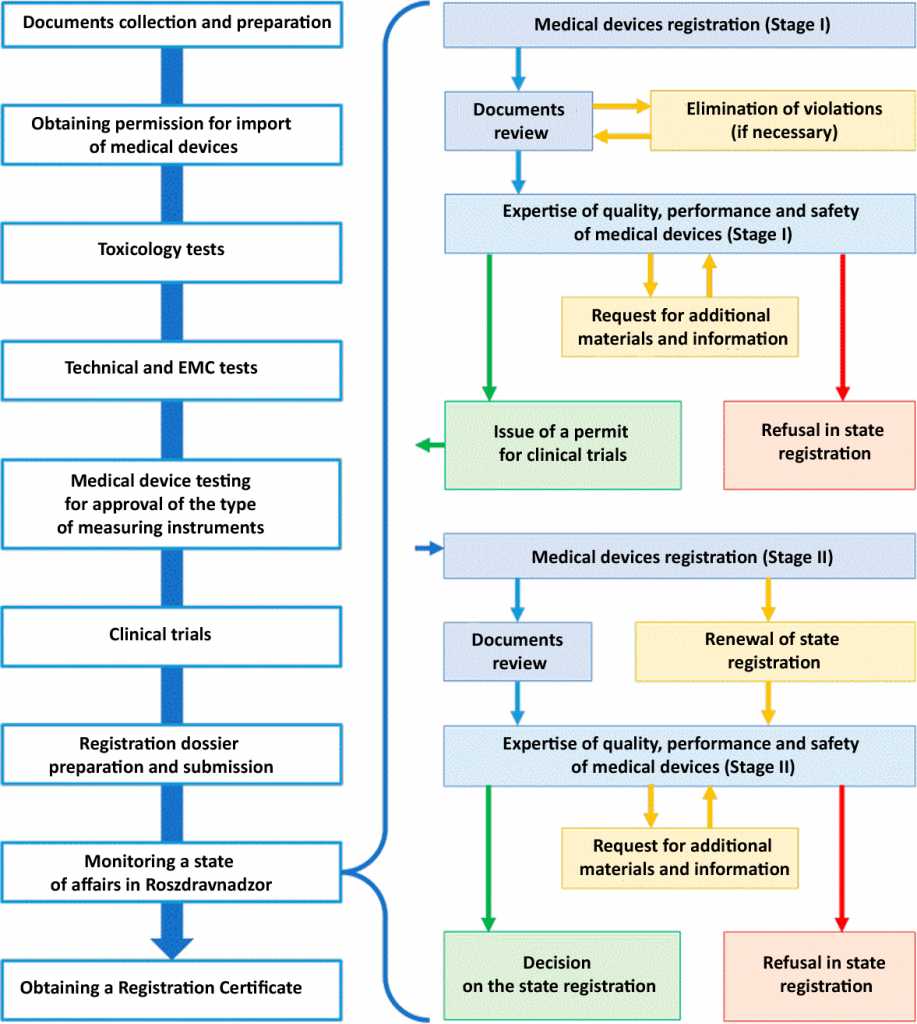
Scheme of the registration process:
Also, SOYUZEKSPERTIZA of the RF CCI renders full range of services for amending the registration dossier (RDA) /
amending the registration certificate (RCA), obtaining duplicates of registration certificates, changing the forms of registration
certificates.
RDA is effected to update information on a medical device in the archive of Roszdravnadzor in case of any changes in the technical
parameters and characteristics of a device. Roszdravnadzor conducts continuous monitoring of compliance of distributed products
and available technical documentation. Any discrepancy leads to suspension of product realization.
RCA is effected in case of any change of information entered into the registration certificate form. Namely, in case of:
- reorganization of a legal entity;
- changing its name (full and shortened (if any), including the brand name), address (location);
- changing the surname, name and patronymic (if available), place of residence of the individual entrepreneur, the
- document proving his/her identity.
- change of address (place of production) of a medical device;
- changing the name of a medical device (in case the properties and characteristics affecting the quality, efficiency
- and safety of a medical device remain unchanged);
- changing the data of legal entity to whose name a registration certificate may be issued, including
- changing its name (full and shortened (if any), including the brand name), address (location);
- reorganization of a legal entity.
- indicating the type of a medical device according to nomenclature classification (in case of its absence).
In case of loss of the Registration Certificate or its damage a duplicate is issued.
Registration Certificate replacement is relevant for certificates valid for an indefinite term, issued before January 1,
2013. The service is available until 2021.
ORDER A FREE REQUEST PROCCESSING
Declaration of Conformity in GOST system. It is a mandatory document for the customs clearance and sale of medical
devices in the territory of the Russian Federation. The Declaration of Conformity is issued according to approved
structure, but does not have a special form (in contrast to the GOST R certificate), instead it is issued on an ordinary
sheet and registered by an accredited certification body.
declaring conformity is marked with a symbol

effective, breakeven and comfortable business. Our experience allows to conduct all procedures as quickly as
possible at the highest professional level.

